Why Your 510k Got a Deficiency Letter — and What to Do About It.
A diagnostic framework for MedTech manufacturers facing a Food and Drug Administration (FDA) deficiency letter in their Human Factors and usability submission for 510k clearance, and what to do about it.
You’ve spent months — perhaps years — developing your medical device. The engineering is sound. The clinical data is solid. You’ve run your summative validation study and prepared your 510k submission. And then the FDA issues a 510k deficiency letter. For many MedTech teams, this is the moment the project grinds to a halt. The expectation of FDA clearance is replaced by confusion, frustration, and a very uncomfortable question: What went wrong?
At THRIVE, we have found that in most cases, something went wrong long before the 510k submission was filed. In this article, we set out to explain why 510k submissions — specifically, why those involving Human Factors and usability evidence — receive 510k deficiency letters, what the most common root causes are, and how to systematically diagnose your situation before jumping into expensive and potentially misdirected remediation efforts.
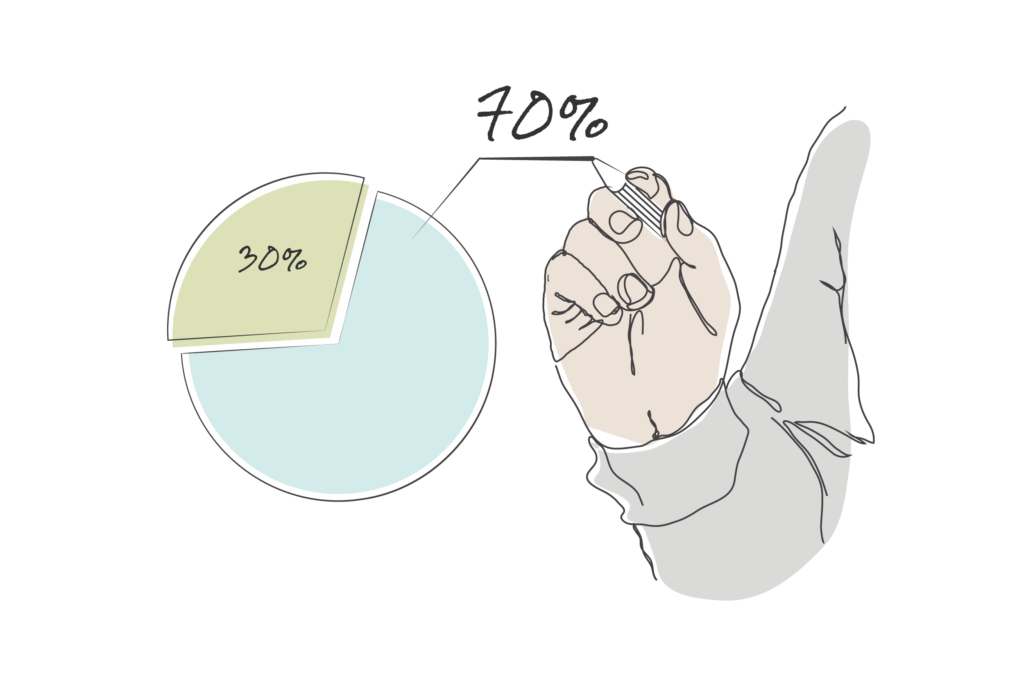
The scale of the problem is significant. According to FDA data, approximately 70% of 510k submissions receive a 510k deficiency letter or some form of request for Additional Information Needed (AINN) on their first cycle of the review process — a figure that has been trending upward since FY2023.[1] The picture is even starker when it comes to integrating Human Factors into medical device development: industry analysis shows that around 90% of human factors testing reports submitted with 510k applications are considered deficient by the FDA.[2] These are not simply a handful of outlying cases. This is the rule, not the exception.
The Root Causes: Why Human Factors Submissions Fail
We have found that FDA 510k deficiency letters in Human Factors are rarely caused by a single, isolated failure. They almost always reflect a systemic gap — one that has been compounded across multiple documents and decisions throughout the medical device design and development process. Our experience with FDA requests and 510k deficiency letters shows that the root causes tend to cluster around five recurring patterns.
1. An Incomplete or Poorly Structured URRA
The Use-Related Risk Analysis (URRA) — an assessment that identifies how device use could lead to harm and ensures risks are minimized — serves as the analytical basis for any Human Factors submission.[3] It is the document that links hazardous-use situations (where incorrect use could cause harm) to patient harm, identifies which tasks are safety-critical, and establishes the rationale for the entire summative study design (the final test to demonstrate the device is safe and effective for users).
An URRA that simply lists possible errors without tracing them to harm severity (the seriousness of potential injury), probability of occurrence (how likely the error is to happen), and risk control measures (steps to reduce risks) is insufficient for a complex device. The FDA expects a document that is comprehensive and traceable to regulatory requirements, such as those outlined in 21 CFR (the Code of Federal Regulations, Title 21, for food and drugs).[4]
2. Summative Study Design That Doesn’t Represent Real-World Use
The FDA’s 2016 Human Factors Guidance (official FDA instructions for designing user studies) is explicit: summative validation studies (final evaluation of the device with users) must simulate actual use conditions with representative users in representative environments that match the device’s intended use.[5] Submissions fail when participant groups don’t reflect the full range of intended users; when scenarios are conducted in conditions that are too controlled, too clean, or too coached; or when pass/fail criteria are defined in ways that don’t correspond to genuine patient risk, or when training decay (allowing time between instruction and testing to simulate real-world memory loss) is insufficient.
For complex devices — particularly robotic surgical systems or devices used in high-pressure clinical environments — the bar for simulation fidelity is high.[6] FDA reviewers will scrutinize whether the study truly challenged the device’s interface in actual use. A summative usability study, designed to “pass” rather than genuinely interrogate the design, is likely to fail FDA review.
3. Unexplained or Inadequately Mitigated Use Errors
Use errors observed during a summative usability study are not automatically disqualifying — but they must be fully explained and adequately mitigated. An FDA submission that logs use errors in data sheets without providing a clear, evidence-based account of why those errors occurred, what their potential harm was, and why they don’t pose an unacceptable risk will attract scrutiny of the testing data. FDA reviewers are experienced at identifying when explanations are superficial or post hoc rationalizations rather than genuine analysis grounded in the URRA and risk framework, conducted at the time of performance testing.[5]
4. Moderation Failures That Compromise Study Validity
How a summative usability study (a final user test) is moderated (guided by a facilitator) matters enormously. If moderators coach participants, provide unsolicited guidance, intervene in ways not specified in the protocol (the formal set of study instructions), or present tasks inconsistently across participant groups, the study’s validity is compromised.[5] This can be difficult for teams to identify themselves — especially when the moderation seems natural and helpful in the moment — but FDA reviewers with access to session videos will identify it quickly.
5. Cross-Document Inconsistency
Perhaps the most insidious root cause in testing data is the disconnect between documents that should be a coherent, integrated evidence package. When the URRA identifies a set of critical tasks, but the protocol tests a different set; when the moderation guide instructs facilitators to observe things that data sheets don’t capture; when the submission narrative describes study conditions that the session videos don’t corroborate — the FDA reviewer is faced with an evidence package that doesn’t hang together. These cross-document inconsistencies are frequently the underlying cause of deficiency notices, even when no single document appears obviously deficient in isolation.[7]
A Diagnostic Framework: Before You Do Anything Else
If you’ve received a 510k deficiency letter, the natural impulse is to start fixing things immediately. Resist it. Jumping directly into remediation — commissioning new studies, revising protocols, rewriting the URRA — without first understanding the root cause of your deficiency carries serious risk. You may address the FDA’s stated points while missing the unstated ones. You may over-invest in areas that don’t need it and under-invest in the areas that do. You may run a new summative study that repeats the same design errors as in the first, thereby demanding additional testing.
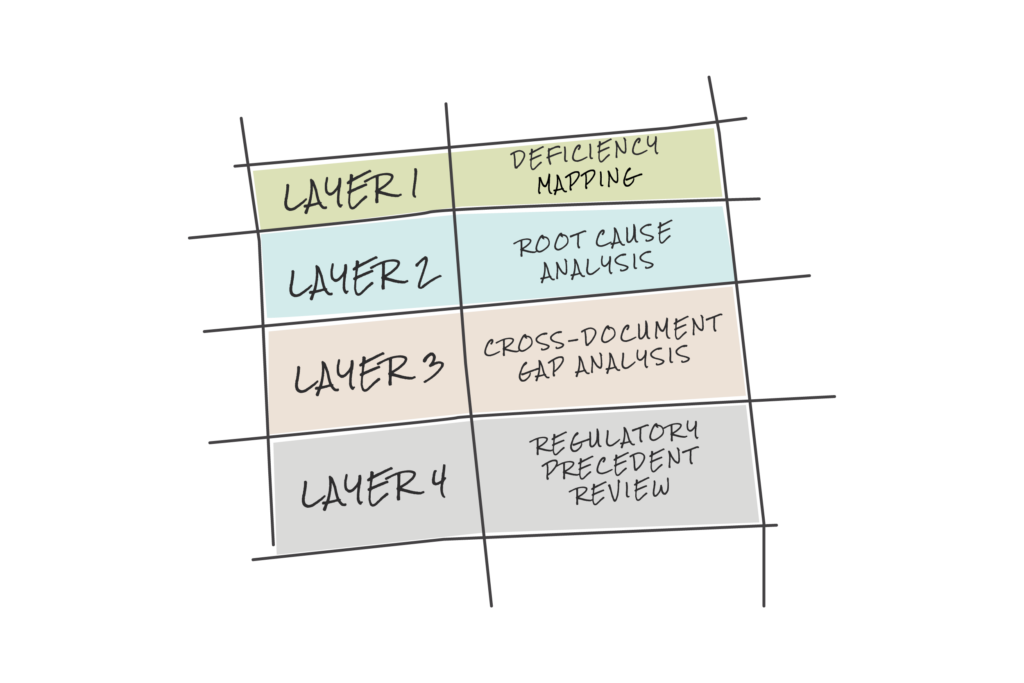
A structured diagnostic process — what we call a Phase Zero approach — is the right first step. It moves through four analytical layers:
1. Deficiency Mapping: Begin with a rigorous, line-by-line analysis of the FDA 510k deficiency letter. Categorize each item by type — study design, participant recruitment, task identification, URRA gap, moderation failure, documentation failure, or data interpretation error. Map every deficiency to the specific FDA guidance clause it references. Build a structured register that will serve as the backbone for everything that follows. Don’t conflate deficiencies that look similar on the surface; they may have different root causes.
2. Root Cause Analysis: For each deficiency, trace backward through the submission materials to identify the source of the failure. Was the URRA missing a user group? Did the protocol fail to derive its critical tasks from the URRA? Did you have the right training decay time? Were pass/fail criteria defined without reference to harm severity? This layer requires access to all submission documents and the discipline to follow the chain of evidence rather than accept surface-level explanations.
3. Cross-Document Gap Analysis: Examine the five core materials — the deficiency notice, the summative validation protocol (the detailed plan for the final user study), the URRA, the data sheets and session videos (records of what happened during the study), and the moderation guide (instructions for study facilitators) — as an integrated evidence package. Map what each document says against every other document. Identify the specific points where they are inconsistent, where linkages are missing, and where documentation has failed to capture what actually happened in the study.
4. Regulatory Precedent Review: Assess your findings against publicly available FDA 510k summaries for comparable devices, published FDA guidance, and known warning letter precedents. This establishes the standard of evidence the FDA has accepted for similar device types and the bar for remediation. You can’t set the target without knowing where it is.
The output of this diagnostic process is not a list of problems, but a clear understanding of the FDA issues. It is a clear, evidence-based picture of what the FDA found, why it found it, and where in the submission the root causes live. Only at this point are you ready to build a remediation plan with genuine confidence.
Building Your Remediation Action Plan
A well-organized remediation plan has three essential characteristics: it is prioritized, sequenced, and clearly scoped. Prioritization means distinguishing between deficiencies that will block clearance entirely and those that, while real, represent lower-order documentation issues. Sequencing means understanding which activities are dependent on others — you cannot design a new summative study before you’ve revised the URRA, and you cannot revise the URRA before you’ve completed the root cause analysis. Scoping means being precise about what needs to change and what doesn’t; over-remediation is as wasteful as under-remediation.
Concretely, a remediation action plan ought to address the following in order:
1. Resolve all URRA gaps identified in the diagnostic, guaranteeing every critical task is identified, every harm pathway is documented, and every risk control is traceable into the study design.
2. Redesign the summative validation protocol to address participant representativeness, scenario fidelity, and pass/fail criteria, in line with the updated URRA.
3. Revise moderation guides to eliminate prompting, coaching, or inconsistent task presentation risks.
4. Conduct new or supplementary usability testing only where the diagnostic has confirmed that existing data cannot be defended, not as a default response to every deficiency.
5. Draft FDA response correspondence that maps each remediation activity directly to each deficiency item, using the deficiency register as the structural backbone
Why Human Factors Expertise Is Non-Negotiable
There is a persistent temptation among MedTech and Pharma manufacturers to treat Human Factors as a documentation exercise — something to be handled late in development by regulatory writers, rather than a rigorous applied science that directs device design from the outset. The deficiency statistics should decisively dispel that view.
The FDA’s expectations in this area have matured significantly. The 2016 Human Factors Guidance established a detailed framework, and FDA reviewers have become increasingly proficient at distinguishing between submissions that really engage with human factors principles and those that treat them as a checkbox. Average 510k submissions now exceed 1,000 pages — double the volume of a decade ago — and FDA reviewers are scrutinizing the human factors elements with corresponding rigor.[8]
Retaining qualified Human Factors experts — people with direct experience in FDA submission requirements, summative study design, URRA methodology, and usability engineering — at the right points in development is not a luxury. It is the single most effective risk mitigation strategy available. An expert who reviews your URRA before the summative study is designed to catch the gaps that would otherwise become FDA deficiencies. An expert who designs your summative protocol against FDA guidance will ensure your study collects the evidence the FDA needs to see. An expert who reviews your session videos independently prior to submission will identify the issues that a deficiency notice would later raise.
When a 510k deficiency letter or an additional information request has already been received, expert involvement becomes even more critical. The diagnostic and remediation process described in this article requires deep familiarity with the FDA’s framework, regulatory precedent, and the specific failure modes that characterize Human Factors deficiencies. Getting this wrong — treating a second submission with the same approach that failed the first — risks a second deficiency cycle, extended market delay, and significantly increased cost.
The Path Forward
A 510k deficiency letter is not the end of the road. Thousands of devices have navigated this process and reached market clearance — including those requiring a De Novo classification — but those that do so efficiently approach remediation systematically rather than reactively. They diagnose before they act. They understand the root causes before designing a remediation plan. They engage experts who can see the evidence as the FDA does.
If your submission has received a Human Factors 510k deficiency letter, the immediate priority is clarity: a clear-eyed understanding of what the FDA found, why it found it, and what needs to happen next. Everything else — the study redesign, the URRA revision, the response correspondence — flows from that foundation.
At THRIVE, we work with MedTech manufacturers to build the human factors evidence the FDA needs — from early URRA development through summative study design, execution, and submission. If you’re facing a 510k deficiency notice and need expert guidance on what it means and how to respond, we’re here to help.
______________________________________________________________________________
References
1. Mastering Responses to FDA 510(k) Additional Information Letters: A Strategic Approach (2023)
2. Human Factors Archives — Industry Analysis of 510(k) Deficiency Rates (2023)
3. FDA: Purpose and Content of Use-Related Risk Analyses for Drugs, Biological Products, and Combination Products (2024)
4. FDA: Content of a 510(k) (2023)
5. FDA: Applying Human Factors and Usability Engineering to Medical Devices (2016)
6. FDA: Safety and Performance-Based Pathway (2023)
7. FDA: Developing and Responding to Deficiencies in Accordance with the Least Burdensome Provisions (2022)
8. MedTech Dive: PMA Major Deficiency Letters Tick Up, 510(k) Asks for Info Flatline: MDUFA Report (2023)
9. FDA: Content of Human Factors Information in Medical Device Marketing Submissions — Draft Guidance (2022)
10. FDA CDRH 2024 Annual Report — Emergo by UL Summary
11. Greenlight Guru: Ready to Submit Your FDA 510(k)? But Wait, There’s No Human Factors!
12. THRIVE: Use-Related Risk Analysis (URRA) for Enhanced Risk Mitigation and Usability
13. THRIVE: URRA — Use-Related Risk Analysis
14, THRIVE: FDA Risk in Medical Device R&D
______________________________________________________________________________
ABOUT THRIVE
THRIVE’s Human Factors professionals have decades of experience applying Human Factors to products ranging from medical devices used by specialized healthcare professionals in clinical environments to combination products used by laypeople in the home.
If it’s your first time applying Human Factors and you need a comprehensive end-to-end Human Factors program, we’ll scope out the program and conduct the activities on your behalf, leaving you time and resources to focus elsewhere. If you’re a resource constrained HFE professional, we’ll step in, providing the teamwork, collaboration, and support to help you meet your goals. Or, if you just want a final sanity check to ensure you’ve met the latest and greatest expectations, we’ll do that too.
Contact us today at 404.228.7342 or email [email protected] to get the conversation started.